Snelheid

Transparantie

Kwaliteit

Snelheid
Wetenschappelijk onderzoek hoeft volgens GPRI niet altijd een jarenlang en complex traject te zijn. Door jarenlange ervaring in het veld, multidisciplinaire teams en transparante tijdslijnen garanderen wij snelheid binnen onze projecten. De deelnemende partijen nemen wij mee in de planning van een studie zodat iedereen weet wat op welke momenten van hen verwacht wordt.
Transparantie
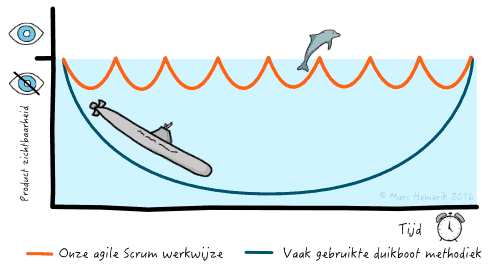
GPRI is een Agile organisatie. Wij gebruiken de Scrum methode om effectiever en flexibeler te werk te gaan. Als deel van de Scrum methode werken wij in korte cycli met regelmatige feedback momenten. Hierdoor is gedurende het hele onderzoek voor alle betrokken partijen duidelijk waar we staan, hoe ver het onderzoek is en of er knelpunten zijn. Zoals weergegeven in de afbeelding, duiken wij niet onder als een duikboot maar komen wij regelmatig aan de oppervlakte zoals een dolfijn om de voortgang te laten zien.
Kwaliteit
Aan wetenschappelijk onderzoek worden hoge kwaliteitseisen gesteld. Voor elk onderzoek wordt een protocol opgesteld die getoetst wordt door de Medisch Ethische Toetsingscommissie (METC), dit is een overheidsinstantie die beoordeelt of wetenschappelijk onderzoek onder de Wet Medisch-wetenschappelijk Onderzoek met mensen (WMO) valt. Als dat het geval is, dan controleert de commissie of de rechten, veiligheid en het welzijn van de proefpersonen die deelnemen aan dit onderzoek worden gewaarborgd. Bij toestemming van de METC kan de praktische uitvoer van het onderzoek beginnen. We verzamelen de benodigde partners en verzorgen training voor alle onderzoekers die het onderzoek gaan uitvoeren.
Alle collega’s binnen GPRI die betrokken zijn bij de uitvoer van wetenschappelijk onderzoek zijn Good Clinical Practice (GCP) getraind en werken volgens de Verklaring van Helsinki, waarin richtlijnen en basisprincipes gegeven worden voor het verrichten van wetenschappelijk geneeskundig onderzoek op mensen en de omstandigheden waaronder deze zijn toegestaan.

















